Aqua regia
Appearance
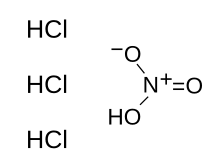
| |
| Names | |
|---|---|
| IUPAC name
nitric acid hydrochloride
| |
| Ither names
aqua regis, nitrohydrochloric acid
| |
| Identifiers | |
3D model (JSmol)
|
|
PubChem CID
|
|
| |
| Properties | |
| HNO3+3 HCl | |
| Appearance | reid, yellae or gowd fumin liquid |
| Density | 1.01–1.21 g/cm3 |
| Meltin pynt | −42 °C (−44 °F; 231 K) |
| Bylin pynt | 108 °C (226 °F; 381 K) |
| miscible in watter | |
| Vapour pressur | 21 mbar |
| Hazards | |
| NFPA 704 | |
Except whaur itherwise notit, data are gien for materials in thair staundart state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Aqua regia (Laitin an Auncient Italian, lit. "regal watter"), aqua regis (Laitin, lit. "king's watter"), or nitro-hydrochloric acid is a heichly corrosive mixtur o acids, a fumin yellae or reid solution. The mixtur is furmed bi freshly mixin concentratit nitric acid an hydrochloric acid,[1] optimally in a vollum ratio o 1:3. It wis named sae acause it can dissolve the sae-cried ryal or noble metals, gowd an platinum. Houiver, titanium, iridium, ruthenium, tantalum, osmium, rhodium an a few ither metals are capable o wistandin its corrosive properties.[2]
Notes
[eedit | eedit soorce]- ↑ The information in the infobox is anly accurate if the molar ratio o nitric acid tae hydrochloric acid is 1:3.
References
[eedit | eedit soorce]- ↑ The acid concentrations in water differ, and indicative values could be 65% w/v for nitric acid and 35% w/v for hydrochloric acid — that is, the actual HNO3:HCl mass ratio is less than 1:2,
- ↑ Encyclopædia Britannica Online. "Aqua regia".

