Glycerol
Appearance
| |||
| |||
| Names | |||
|---|---|---|---|
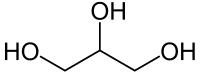
| IUPAC name
propane-1,2,3-triol
| |||
| Ither names
glycerin
glycerine propanetriol | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties | |||
| C3H8O3 | |||
| Molar mass | 92.09 g·mol−1 | ||
| Appearance | colorless liquid hygroscopic | ||
| Odour | odorless | ||
| Density | 1.261 g/cm3 | ||
| Meltin pynt | 17.8 °C (64.0 °F; 290.9 K) | ||
| Bylin pynt | 290 °C (554 °F; 563 K) | ||
| Refractive index (nD) | 1.4746 | ||
| Viscosity | 1.412 Pa·s[1] | ||
| Hazards | |||
| NFPA 704 | |||
| Flash pynt | 160 °C (320 °F) (closed cup) 176 °C (349 °F) (open cup) | ||
Except whaur itherwise notit, data are gien for materials in thair staundart state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Glycerol (or glycerine, glycerin) is a simple polyol (succar alcohol) compoond. It is a colorless, odorless, viscous liquid that is widely uised in pharmaceutical formulations. Glycerol haes three hydroxyl groups that are responsible for its solubility in watter an its hygroscopic naitur. The glycerol backbone is central tae aw lipids kent as triglycerides. Glycerol is sweet-tastin an o law toxicity.
References
[eedit | eedit soorce]- ↑ "Viscosity of Glycerol and its Aqueous Solutions". Retrieved 19 Apryle 2011.
- ↑ Lide, D. R., ed. (1994). CRC Handbook of Data on Organic Compounds (3rd ed.). Boca Raton, FL: CRC Press. p. 4386.