Paracetamol
[1] Archived 2018-10-11 at the Wayback Machine
 | |
 | |
| Clinical data | |
|---|---|
| Tred names | Tylenol (USA), Anacin Aspirin Free (USA), Apra (USA), Crocin (Indie), Feverall (USA), Genapap (USA), Panadol (UK), Panadol (Australie), Panodil (Denmark), Efferalgan (Fraunce), Doliprane (Fraunce) Dafalgan (Fraunce) Alvedon (Swaden) |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a681004 |
| Leecence data |
|
| Pregnancy category | |
| Routes o admeenistration | Oral, rectal, intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ~100% |
| Metabolism | 90 tae 95% Hepatic |
| Biological hauf-life | 1–4 h |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Nummer | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.002.870 |
| Chemical and physical data | |
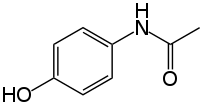
| Formula | C8H9NO2 |
| Molar mass | 151.17 g/mol |
| 3D model (Jmol) | |
| Density | 1.263 g/cm3 |
| Meltin pynt | 169 °C (336 °F) [2][3] |
| Solubility in watter | 12.78[1] mg/mL (20 °C) |
| |
| |
| (verify) | |
Paracetamol INN (/ˌpærəˈsiːtəmɒl/ or /ˌpærəˈsɛtəmɒl/), or acetaminophen USAN ![]() i/əˌsiːtəˈmɪnəfᵻn/, chemically named N-acetyl-p-aminophenol, is a widely uised ower-the-coonter analgesic (pain reliever) an antipyretic (fiver reducer).
i/əˌsiːtəˈmɪnəfᵻn/, chemically named N-acetyl-p-aminophenol, is a widely uised ower-the-coonter analgesic (pain reliever) an antipyretic (fiver reducer).
Paracetamol is clessifee'd as a mild analgesic. It is commonly uised for the relief o heidaches an ither minor aches an pains an is a major ingredient in numerous cauld an flu remedies. In combination wi opioid analgesics, paracetamol can an aa be uised in the management o mair severe pain sic as post-surgical pain an providin palliative care in advanced cancer patients.[4] Tho acetaminophen is uised tae treat inflammatory pain, it is nae generally clessifee'd as an NSAID acause it exhibits anly waik anti-inflammatory activity.
The onset o analgesia is approximately 11-29.5 minutes efter oral admeenistration o paracetamol, an its hauf-life is 1–4 oors. While generally safe for uise at recommendit doses (1,000 mg per single dose an up tae 4,000 mg per day for adults),[5] acute owerdoses o paracetamol can cause potentially fatal neer, brain an liver damage an, in rare individuals, a normal dose can dae the same. The risk mey be hichtened bi chronic alcohol abuse. Paracetamol toxicity Archived 2018-10-11 at the Wayback Machine is the foremost cause o acute liver failure in the Wastren warld, an accoonts for maist drug owerdoses in the Unitit States, the Unitit Kinrick, Australie an New Zealand.[6][7][8][9]
It is the active metabolite o the coal tar–derived phenacetin, ance popular as an analgesic an antipyretic in its awn richt. Houiver, unlik phenacetin an its combinations, paracetamol is nae considered carcinogenic at therapeutic doses.[10] The wirds acetaminophen (uised in the Unitit States,[11] Canadae, Japan, Sooth Korea, Hong Kong, an Iran) an paracetamol (uised elsewhaur) baith come frae a chemical name for the compoond: para-acetylaminophenol an para-acetylaminophenol. In some contexts, it is simply abbreviated as APAP, for acetyl-para-aminophenol.
References
[eedit | eedit soorce]- ↑ Granberg RA, Rasmuson AC (1999). "Solubility of paracetamol in pure solvents". Journal of Chemical & Engineering Data. 44 (6): 1391–95. doi:10.1021/je990124v.
- ↑ Karthikeyan, M.; Glen, R. C.; Bender, A. (2005). "General Melting Point Prediction Based on a Diverse Compound Data Set and Artificial Neural Networks". Journal of Chemical Information and Modeling. 45 (3): 581–590. doi:10.1021/ci0500132. PMID 15921448.
- ↑ "melting point data for paracetamol". Lxsrv7.oru.edu. Archived frae the original on 30 Juin 2012. Retrieved 19 Mairch 2011.
- ↑ Scottish Intercollegiate Guidelines Network (SIGN) (2008). "6.1 and 7.1.1". Guideline 106: Control of pain in adults with cancer (PDF). Scotland: National Health Service (NHS). ISBN 978 1 905813 38 4.
- ↑ Acetaminophen Drugs.com
- ↑ Daly FF, Fountain JS, Murray L, Graudins A, Buckley NA (2008). "Guidelines for the management of paracetamol poisoning in Australia and New Zealand—explanation and elaboration. A consensus statement from clinical toxicologists consulting to the Australasian poisons information centres". Med J Aust. 188 (5): 296–301. PMID 18312195.CS1 maint: multiple names: authors leet (link)
- ↑ Khashab M, Tector AJ, Kwo PY (2007). "Epidemiology of acute liver failure". Curr Gastroenterol Rep. 9 (1): 66–73. doi:10.1007/s11894-008-0023-x. PMID 17335680.CS1 maint: multiple names: authors leet (link)
- ↑ Hawkins LC, Edwards JN, Dargan PI (2007). "Impact of restricting paracetamol pack sizes on paracetamol poisoning in the United Kingdom: a review of the literature". Drug Saf. 30 (6): 465–79. doi:10.2165/00002018-200730060-00002. PMID 17536874.CS1 maint: multiple names: authors leet (link)
- ↑ Larson AM, Polson J, Fontana RJ, et al. (2005). "Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study". Hepatology. 42 (6): 1364–72. doi:10.1002/hep.20948. PMID 16317692.CS1 maint: multiple names: authors leet (link)
- ↑ Bergman K, Müller L, Teigen SW (1996). "The genotoxicity and carcinogenicity of paracetamol: a regulatory (re)view". Mutat Res. 349 (2): 263–88. doi:10.1016/0027-5107(95)00185-9. PMID 8600357.CS1 maint: multiple names: authors leet (link)
- ↑ Bradley, N (1996). "BMJ should use "paracetamol" instead of "acetaminophen" in its index". BMJ. 313 (7058): 689. PMC 2351967. PMID 8811774.
Freemit airtins
[eedit | eedit soorce]- Paracetamol at Chemsynthesis
- Paracetamol Information Centre Archived 2006-09-25 at the Wayback Machine
- Paracetamol Internaitional Chemical Safety Cards
- The Julius Axelrod Papers
- FDA: Safe Uise o Ower-the-Coonter Pain Relievers/Fever Reducers
- FDA: Consumer Update "Acetaminophen and Liver Injury: Q and A for Consumers" (link) Archived 2013-07-19 at the Wayback Machine
- FDA: Consumer Update "Acetaminophen and Liver Injury: Q and A for Consumers" (PDF) Archived 2013-09-19 at the Wayback Machine
- U.S. Naitional Library o Medicine: Drug Information Portal–Paracetamol
- Acetaminophen boond tae proteins in the PDB
